Biological Inactivation
Low temperature plasma sterilization technology for prevent infection accidents
Low Temperature Plasma Sterilizer Technology
Biological Inactivation
Plasmapp's prevention solution is the world's fastest low temperature sterilization solution using direct sterilant injection technology
using an impermeable pouch, which can maximize usage efficiency while preventing thermal damage to sophisticated medical devices.
By using Plasmapp's differentiated plasma technology, user safety is secured in the sterilization process.
By using Plasmapp's differentiated plasma technology, user safety is secured in the sterilization process.
Pouch Mode
7 minutes fast sterilization with the world's first direct injection pouch and patented sterilization technology
7 minutes fast sterilization with the world's first direct injection pouch and patented sterilization technology
Vacuum sealing pouches enable staff to have an immediate check on the sterile condition of the sterilized tools inside
Safe disposable sterilant cassettes
Compatible with most medical devices and materials that are heat and moisture intolerable
Simple to operate, install, and monitor (only requires electrical outlet)
Novel sterile packaging to enable super-fast 7 min sterilization cycle
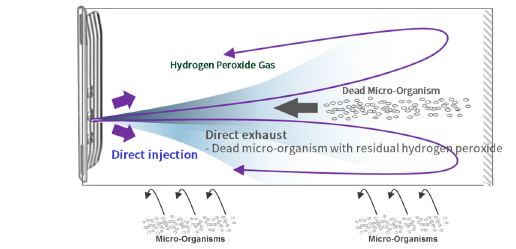
- Direct sterilant injection enhances sterilization efficiency.
- Direct vacuum pumping shortens purification process time.
- Independent pressure control allows unique sterilization cycle including compression process.
- Compression maximizes sterilization efficiency to obtain 7 min cycle.

- Impermeable pouch allows direct sterilization with compression process and vacuum sealing after the cycle.
- Vacuum sealing visualizes sterile condition which extends shelf-life of sterilized devices and provides advanced infection control solution.
* Pouch sterilization is only available for STERLINK FPS-15s Plus and STERLINK mini.
STERLINK sterilization system for the most effective surgery
The world's first small plasma sterilizer approved by the US FDA



510(K) clearance for STERLINK
sterilizer system of Plasmapp
(K220345 / K212200 / K212193 / K212198)
sterilizer system of Plasmapp
(K220345 / K212200 / K212193 / K212198)

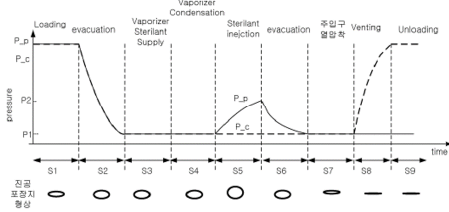
Sterilization Performance
STERLINK verifies sterilization performance through the following lumen tests
Single-channel lumen claims for STERLINK
Single-channel lumen claims for STERLINK
- Ø0.7 x 500mm Stainless steel
- Ø2.0 x 1,500mm Stainless steel
- Ø1.0 x 2,000mm PTFE
STERLINK - inside diameter 1mm and a length up to 2,000mm
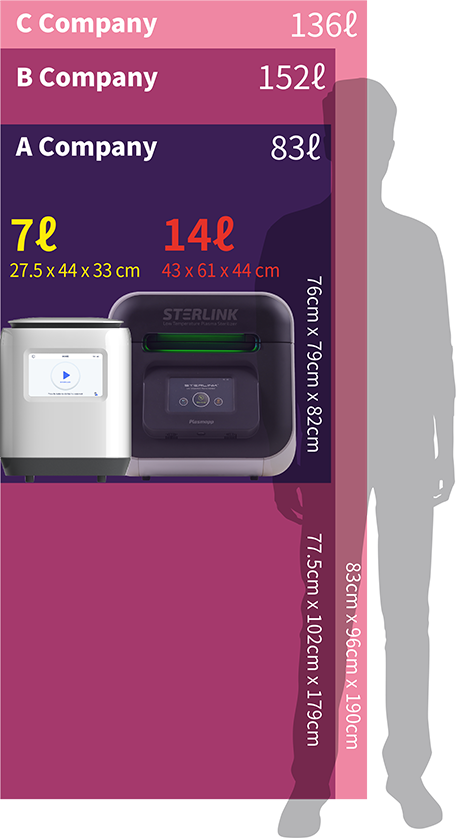
Competitor A
length up to 500mm
length up to 500mm
Competitor B
length up to 1,000mm
length up to 1,000mm
Plasmapp
STERLINK
STERLINK
Innovative plasma sterilization technology verified
through patents and research papers
through patents and research papers
Forced convective heating for low-temperature sterilization
Publication expected in 2021 (AIP) - https://doi.org/10.1063/5.0048688
Review of Scientific Instruments 92, 064902 (2021) conducted by Dr. Youbong Lim, Dr. Wonho Choe, Dr. Seung Hun Lee, Dr. Jun Young Kim and Dr. Hyun Jeong Jeon
Publication expected in 2021 (AIP) - https://doi.org/10.1063/5.0048688
Review of Scientific Instruments 92, 064902 (2021) conducted by Dr. Youbong Lim, Dr. Wonho Choe, Dr. Seung Hun Lee, Dr. Jun Young Kim and Dr. Hyun Jeong Jeon
WE WOULD LOVE TO HEAR ABOUT
YOUR INTEREST IN PRODUCTS
YOUR INTEREST IN PRODUCTS